Saltar al contenido | | A partnership between Sanofi Pasteur and FISABIO in Valencia Spain has been established. FISABIO is in charge to coordinate and animate the GIHSN network. This partnership is mainly focused on Capacity building workshops organization; Facilitation of scientific exchanges and trouble shooting between the network members; Facilitation and execution of pooled-analyses; and Organization of a yearly workshop to share and discuss results.
The coordinator and a member of the coordination team as epidemiologists, work and travel to global sites to help the partners in trouble shooting and quality control.
Sanofi Pasteur has agreed to provide funding, and solely be present in terms of providing support to the different parties. Study sites databases used for the pool analysis are owned by the study site. The data is shared with the FISABIO to allow for the pooled analysis.
Study site's anonymous datasets are shared with FISABIO (one month after the beginning of the study site activities, at the end of the second month and at the end of the influenza season for the definitive pooled analysis)
FISABIO is responsible for writing the final report and preparing publications of the study results. There is a Steering Committee who will comment on the results and interpretations of the findings in advance of publication.
A Scientific Committee will collaborate when requested to provide scientific advice and comments to the pooled analysis. Involvement of health authorities and other key experts is strongly welcomed, and with help a strong international visibility (CDC, WHO, GII …) is ensured. 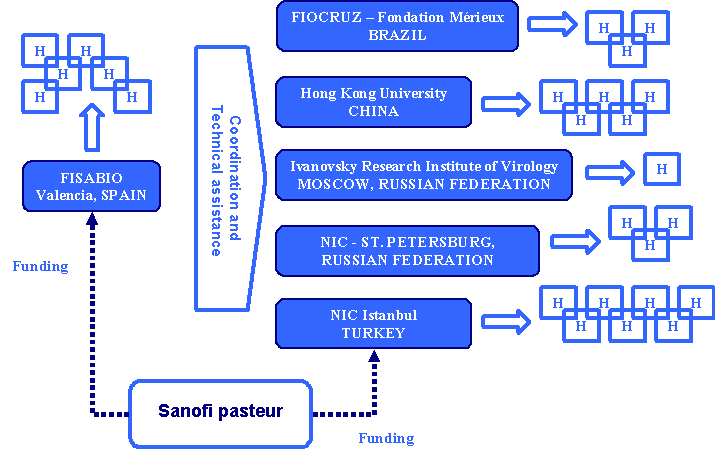 Steering committee:
A group of persons contributing to the management and proceedings of the Study. This group is foreseen for the purpose of providing scientific advice and guidance during the setting up and conduction of the study
The Steering Committee is composed of two epidemiologists in charge of the project from FISABIO, two from Sanofi Pasteur, and each sites' Principal Investigator.
The Steering Committee shall collaborate and contribute to the following missions:
(i) To monitor compliance with the protocol
(ii) To provide epidemiological support and scientific advice
(iii) To provide comments to the pooled analysis plans
(iv) To provide input into discussing and interpreting the pooled analyses study Results
(v) To comment timely on results/interpretations of findings in advance of publication
Scientific Committee:
The Scientific Committee is composed by the Steering Committee, and by each Study Site coordinator of the study and one representative of each of the hospitals involved in the Study, two virologists, one from FISABIO and one for the Study Sites (not counting PIs or coordinators), and two external experts.
The Scientific Committee shall collaborate and contribute to the following missions:
(i) To provide scientific advice.
(ii) To provide comments to the pooled analysis plans.
(iii) To provide input into discussing and interpreting the pooled analyses study Results.
(iv) To comment timely on results/interpretations of findings in advance of publication. | | | | | | | | | | | |